Respiratory failure
The links are provided to make navigation through the module easier. They contain essential information that form a part of this module.
Educational Objectives
By the end of this module you should be able to:
- Understand the pathophysiological mechanisms of respiratory failure
- Recognise the symptoms and signs of respiratory failure
- List the common and uncommon causes of respiratory failure
- Safely prescribe oxygen in respiratory failure
- Perform basic emergency management of patients in respiratory failure
- Identify patients who might benefit from non invasive or invasive ventilatory support
Module 1
Respiratory failure is, by definition, a failure of gas exchange. There are two types:
- Type 1: hypoxaemia with a normal or low CO2
- Type 2: hypoxaemia with a high CO2
The main symptom is shortness of breath.
Signs of respiratory failure
Overall look | The patient is usually pale, sweaty, clammy and looks terrified, exhausted or obtunded Most patients with respiratory failure sit up unless the cause of respiratory failure is CNS related |
Respiratory rate | < 8 or > 30 /min (slow respiratory rate in association with fatigue is an ominous sign) interrupted speech (cannot finish sentence) |
Pulse rate | Tachycardia is usually associated with respiratory failure Bradycardia is usually a late and sinister sign |
SpO2 < 88% | This roughly corresponds to a PaO2 of 8 KPa although the variability is wide |
Confusion, agitation or convulsions | Usually caused by hypoxia and can put the patient and staff at risk of injury |
Reduction in level of consciousness | Usually associated with hypercapnia or severe hypoxia |
Chest signs | Looking at the patient’s chest movements (or lack of) is crucial. Look for:
systematic chest examination is described elsewhere |
CVS | Don’t forget to examine the cardiovascular system as heart failure is a common cause of respiratory failure. |
There are several pathophysiological mechanisms underlying respiratory failure, they are:
- Alveolar hypoventilation
- Diffusion deficit
- Shunt
- Ventilation – perfusion mismatch
We will now discuss all these in order.
Module 2
Hypoventilation
Two things cause alveolar hypoventilation:
1. A reduction in minute ventilation.
Common causes of a reduction in minute ventilation
Lungs e.g. COPD*, bronchospasm |
Thoracic cage e.g. ankylosing spondylitis |
Muscles e.g. myopathy |
Neuromuscular junction e.g. myasthenia gravis |
Nerves e.g. MND |
Spinal Cord e.g. trauma |
Brain Stem e.g. CVA, drugs |
* In COPD hypoventilation occurs due to loss of lung tissue and elasticity. This leads to chest hyperexpansion and the inability to ventilate adequately. Loss of lung tissue also leads to an increase in dead space. In addition, bronchospasm might contribute to the respiratory compromise seen in COPD patients.
2. An increase in the proportion of dead space ventilation which can either be anatomical or physiological.
Anatomical dead space might increase by adding tubing to the breathing circuit |
Physiological dead space might increase when lung tissue is destroyed (COPD) or when perfusion is decreased out of proportion with ventilation (e.g. hypotension causing reduced apical lung perfusion) |
The characteristic of hypoventilation is CO2 retention (Type 2 respiratory failure). PaCO2 is inversely related to alveolar ventilation, see fig 1.
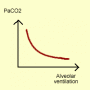
Figure 1. Relationship between PaCO2 and VA (alveolar ventilation)
A rise in PaCO2 in the alveoli leads to an increase in oxygen requirements because the accumulated CO2 molecules displace O2 molecules (see alveolar gas equation below).
Alveolar gas equation
PAO2 is the partial pressure of oxygen in alveolar air. PIO2 is the partial pressure of oxygen in inspired air. PACO2 is partial pressure of CO2 in alveolar air. R is the respiratory quotient. |
Almost every cause of respiratory failure might eventually cause CO2 retention because of respiratory muscle fatigue. Some conditions normally present with Type 1 respiratory failure, but when severe present with marked CO2 retention (e.g. cardiogenic pulmonary oedema). This is because of a profound V/Q mismatch.
Hypoxaemia caused by hypoventilation is easily corrected with low dose oxygen supplementation. If high dose oxygen is required, there is an additional or alternative cause of hypoxaemia.
Module 3
Diffusion deficit
This means that a pathological process is affecting the barrier which is normally present between alveolar gas and the capillary blood. In health, this barrier is very thin and is made of the alveolar epithelial cell, the interstitial space and the capillary endothelium, fig 2.
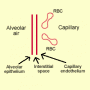
Figure 2a. A schematic representation of the alveolar-capillary unit. The normal diffusion barrier is made of an alveolar epithelial cell, the interstitial space and a capillary endothelial cell.
The most common acute cause is interstitial pulmonary oedema.
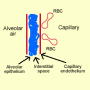
Figure 2b. In pulmonary oedema, fluid accumulates in the interstitial space increasing resistance to gas exchange. In the more advanced stages, fluid accumulates in the alveolus further impeding gas exchange.
The most common chronic cause is pulmonary fibrosis.
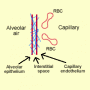
Figure 2c. Pulmonary fibrosis. The diffusion barrier is fibrotic and gas exchange is impaired.
Diffusion deficit from fluid or fibrosis often contributes to respiratory failure in combination with other conditions, but rarely causes it on its own in ICU. When chronic fibrosis causes respiratory failure, patients do not benefit from ventilatory support. Acute fibrosis (e.g. non specific interstitial pneumonitis) presents in a very similar fashion to pneumonia and may respond to immunosuppression.
Module 4
Shunt
Shunt occurs when venous blood mixes with arterial blood either by bypassing the lungs completely (extra-pulmonary shunt) or by passing through the lungs without adequate oxygenation (intra-pulmonary shunt).
Extra-pulmonary
Extra pulmonary (cardiac) shunting is not commonly seen in adult practice. Even when a lesion causes communication between the right and left heart, initially the blood flow will be from left to right. This will cause a reduction in cardiac output and volume overload of the right heart but not shunt. Eventually, compensatory changes may take place, which cause blood to flow from the right to the left heart.
Intra-pulmonary
Shunt occurs when blood is transported through the lungs without taking part in gas exchange. The commonest causes are alveolar filling (with pus, oedema, blood or tumour) and atelectasis, fig 3.
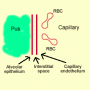
Figure 3. Pneumonia. The alveoli are filled with pus preventing gas exchange. Increasing FiO2 will not improve gas exchange as there is no air in the alveolus.
Increasing FiO2 does not normally correct hypoxia caused by pure shunt. This is because the shunted blood in the diseased alveoli does not come in contact with alveolar gas. The deoxygenated blood leaving the diseased alveoli mixes with blood coming from healthy alveoli. In the relatively healthy alveoli, the oxygen saturation will be around 97-99% regardless of the increase in FiO2. The effect of increasing FiO2 on the blood leaving these alveoli will only be an increase in dissolved oxygen, which contributes little to oxygen delivery to tissues.
Despite this, it is almost always worth trying to increase FiO2, either for the small increase in PO2, or to assess the effect on the other areas of lung where different processes might also be taking place.
Module 5
Ventilation – Perfusion (V/Q) mismatch
V/Q mismatch is the presence of a degree of shunt and a degree of dead space in the same lung. It is a component of most causes of respiratory failure and is the commonest cause of hypoxaemia.
Because of the complicated structure of the lungs, it is impossible to describe this condition in anatomical terms. A patient with this condition is likely to have areas in the lungs that are better perfused than ventilated and areas that are better ventilated than perfused. This occurs in normal lungs to some extent. The difference in V/Q mismatch is that the extent to which this occurs is significantly increased.
Because of the flat upper portion of the Oxyhaemoglobin dissociation curve (fig 4), blood leaving the relatively healthy alveoli will have an oxygen saturation of about 97%. Blood leaving alveoli that do not have optimum V/Q ratios will have a much lower oxygen saturations . The admixture of all the blood leaving the alveoli results low oxygen saturations and hypoxaemia.
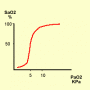
Figure 4. Oxygen haemoglobin dissociation curve. SaO2 = arterial haemoglobin saturations. PaO2 = partial pressure of oxygen.
In general, this cause of respiratory failure responds to oxygen therapy, although the response varies depending on the precise nature and size of the V/Q mismatch.
Module 6
Management of acute respiratory failure
There are two components to the management of respiratory failure: emergency management (resuscitation), and definitive diagnosis and treatment of the underlying condition.
Emergency management
As always, this commences with an assessment of ABCDE. This approach is detailed elsewhere in this induction programme and most doctors will be very familiar with its principles. Make sure the airway is patent and protected, examine the patient and deal with life threatening emergencies. Gain i.v. access. Think about fluid therapy.
How much oxygen?
The simple answer to this is ‘enough’. If the SpO2 is normal, oxygen is not required.
If the SpO2 is low, then high flow oxygen in the short term will do little harm. It should be titrated quickly downwards using SpO2 as a guide. Aim for SpO2 of 92%.
In very few patients (those with clinically severe COPD who have compensated type II respiratory failure – a high bicarbonate with a high CO2) oxygen should be titrated upwards carefully with regular checks of the clinical status (mental state, ventilatory pattern) and blood gases (is CO2 rising?). These patients may hypoventilate when given too much oxygen. This may cause a respiatory arrest in severe circumstances, but more commonly will lead to profound hypoxaemia. This occurs when the FiO2 is reduced following a rise in CO2 (the hypoxia from the reduced FiO2 exacerbated by the high alveolar CO2).
In these patients, aim for SpO2 of 88 –92%. Always assess the response of the patient to your intervention: e.g. has the SpO2 increased, has the CO2 increased?
Definitive diagnosis and treatment
Respiratory failure is NOT an adequate diagnosis. It is a description of a condition that results from many underlying disorders. It is impossible to properly direct further investigation and treatment without a diagnosis. Treatment of the underlying problem is beyond the scope of this review but thought should be given to diseases listed in the table below.
Common | Exacerbation of COPD |
LVF | |
Community Acquired Pneumonia/Hospital Acquired Pneumonia | |
Acute asthma | |
Drug overdose | |
ARDS/ALI | |
Reduced GCS | |
Atelectasis | |
Abdominal splinting | |
Pulmonary embolus | |
Pneumothorax | |
less common | |
Valvular heart disease
Interstitial Lung Disease (e.g. acute interstitial pneumonitis, drug induced pneumonitis, pulmonary fibrosis)
Extrinsic allergic alveolitis
Pulmonary haemorrhage
Organising Pneumonia
Fat emboli
Some causes of respiratory failure.
We will now discuss ARDS further.
Module 7
Acute Respiratory Distress Syndrome (ARDS) & Acute Lung Injury (ALI)
Acute Respiratory Distress Syndrome (ARDS) is under diagnosed. Often the diagnosis is never changed from the underlying condition (pneumonia, aspiration, pancreatitis etc). The diagnostic criteria are listed in the table below.
Acute onset |
Gas exchange abnormaility* |
Bilateral infiltrates on CXR |
Non-cardiogenic in origin |
Known causative condition |
Diagnostic criteria for ARDS/ALI.
*PaO2/FiO2 < 27 KPa for ARDS
PaO2/FiO2 < 40 KPa for ALI
It is the commonest cause of difficulty with ventilation in ICU. It is usually seen as part of a generalised inflammatory response with other organ dysfunction. If the lungs are the only failing organs, be suspicious of another cause of respiratory failure.
Treatment is based on the treatment of the underlying disease. Supportive therapy with ventilation allows time to treat the cause. Ventilation can worsen the inflammatory process and cause further deterioration unless care is taken to avoid ventilator induced lung injury. There are no specific treatments. Diuresis is reasonable, but often difficult to achieve without worsening other organ perfusion.
The underlying conditions that trigger ARDS can be either direct, if they affect the lungs (thoracic ARDS), or indirect (extrathoracic ARDS).
Direct | Pneumonia |
Inhalation or aspiration of toxic substances (e.g. smoke or gastric contents) | |
Contusion | |
Indirect | Sepsis of any cause |
Pancreatitis | |
Burns | |
Profound/prolonged hypotension | |
Multiple transfusions | |
Sometimes both conditions are present, e.g. pneumonia and sepsis | |
Module 8
Indications for ventilation in respiratory failure
This is a difficult decision and should always take place in consultation with a senior doctor. If the patient is causing you or the referring medical or nursing staff significant worry, discuss with your seniors immediately.
Indications for ventilation
| Hypoxaemia |
| Hypercarbia |
| Exhaustion |
| Increase in work of Breathing |
The non-invasive methods of respiratory support described below are useful if the patient is likely to respond quickly (e.g. LVF, acute exacerbation of COPD). If the patient is in respiratory failure because of an illness that is likely to be prolonged (e.g. ARDS, pneumonia) then invasive ventilation is usually more appropriate.
CPAP
This is the application of continuous positive pressure to the patient’s respiratory system. It normally refers to the non invasive application of a tight fitting mask that is connected to an oxygen delivery system which has an outlet valve (or something similar). This valve allows the build up of pressure especially on expiration. The tight fitting mask is essential to maintain the positive pressure. A hood that covers the patient’s head is also in use and can be more comfortable than a face mask. Pressures usually employed are between 5 and 10 cm of H2O. CPAP is usually used to improve oxygenation not to treat hypercarbia. |
Continuous Positive Airway Pressure (CPAP) is an extremely useful technique for some forms of respiratory failure. CPAP systems can deliver high FiO2. CPAP works particularly well for cardiogenic pulmonary oedema and can allow time for medical therapy to work. It may also help in ARDS. The effect is more variable in other forms of respiratory failure.
Non-Invasive Ventilation (NIV)
Patients with hypercapnic, acidotic exacerbations of COPD should be offered Non Invasive Ventilation (NIV) on the medical wards at an early stage in the illness. This reduces mortality and the need for intubation. Most NIV equipment cannot deliver high FIO2.
Think about NIV When called to see any patient with respiratory failure. Beware that if the patient is in distress then NIV is not appropriate. In this case the correct course of action would be to proceed to invasive ventilation as soon as possible. Always discuss with your senior cover.
If the underlying disease is not likely to resolve quickly (e.g. pneumonia) and the patient becomes dependent on NIV, then invasive ventilation is likely to be the best option. This is because this group of patients do not tolerate even short periods without NIV. This will make it difficult to deliver other aspects of patient care e.g. feeding, drinking, mouth care..etc. This is different to COPD patients with acute exacerbations, as they can normally tolerate a few minutes off NIV to eat or drink.
CONTRAINDICATIONS TO NIV |
| Coma (except in CO2 narcosis when a short trial of NIV might be attempted by an experienced clinician) |
| Need for airway protection |
| Agitation/delirium |
| Facial & anatomical abnormalities |
| Recent upper GI surgery with anastomosis |
| Patient refusal |
NIV can also be used as a bridge to invasive ventilation, but always discuss with your senior cover.
